Alongside their native, functional structure, proteins tend to aggregate, resulting in the irreversible loss of the native protein conformation and function.
Protein aggregates can have deleterious if not fatal effects, as manifested in human amyloidal diseases such as Alzheimer (AD) and Parkinson disease (PD). At the molecular level, there is a crucial lack of understanding on how soluble proteins convert to pathological insoluble amyloid aggregates. Recently, liquid-liquid phase separation (LLPS; generating intracellular membraneless compartments in biological cells) was proposed to be a key intermediate for protein aggregation. This M2 project aims at obtaining insights into changes in molecular structure and dynamics during fibrillation of two medically important intrinsically disordered proteins (i.e. tau and -synuclein, involved in AD and PD, respectively) through the application of a broad arsenal of experimental and computational biophysical techniques, including neutron spectroscopy and molecular dynamics simulations. The project is in collaboration with Prof. D. Tobias (UC Irvine), Dr. Yann Fichou (IECB Bordeaux) and Dr. Tilo Seydel (Institut Laue Langevin, Grenoble).
Competences that will be acquired after the intership : Protein expression and purification, neutron scattering
Webpage: See here
Selected publications:
QPounot, K., G. W. Grime, A. Longo, M. Zamponi, D. Noferini, V. Cristiglio, T. Seydel, E. F. Garman, M. Weik, V. Fodera and G. Schiro (2021). "Zinc determines dynamical properties and aggregation kinetics of human insulin." Biophys J 120: 886
Pounot, K., H. Chaaban, V. Fodera, G. Schiro, M. Weik and T. Seydel (2020). "Tracking Internal and Global Diffusive Dynamics During Protein Aggregation by High-Resolution Neutron Spectroscopy." J Phys Chem Lett 11: 6299
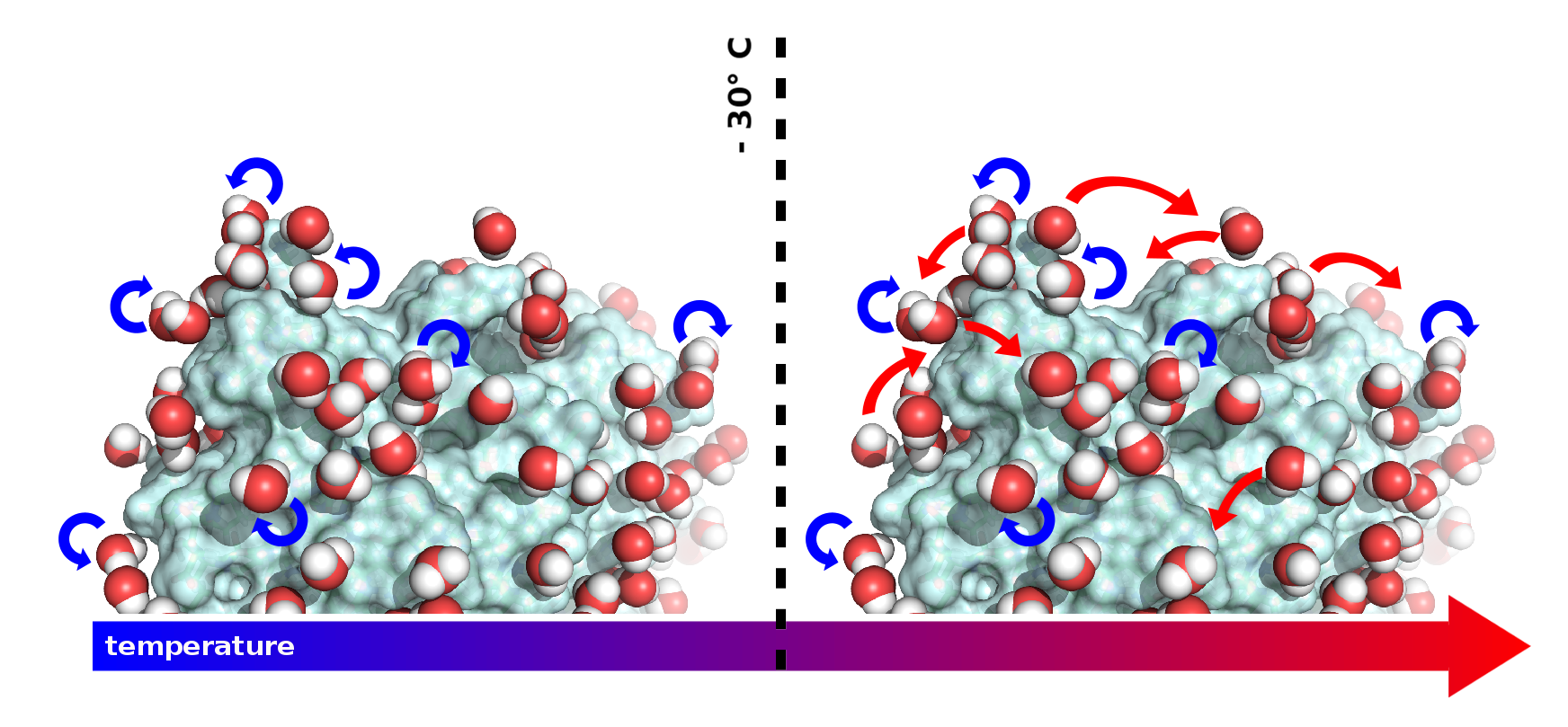
Schiro, G., Y. Fichou, F. X. Gallat, K. Wood, F. Gabel, M. Moulin, M. Hartlein, M. Heyden, J. P. Colletier, A. Orecchini, A. Paciaroni, J. Wuttke, D. J. Tobias and M. Weik (2015). "Translational diffusion of hydration water correlates with functional motions in folded and intrinsically disordered proteins." Nat Commun 6: 6490.
Fichou, Y., G. Schiro, F. X. Gallat, C. Laguri, M. Moulin, J. Combet, M. Zamponi, M. Hartlein, C. Picart, E. Mossou, H. Lortat-Jacob, J. P. Colletier, D. J. Tobias and M. Weik (2015). "Hydration water mobility is enhanced around tau amyloid fibers." Proc Natl Acad Sci U S A 112: 6365